Brii Biosciences Provides Corporate Update and Reports Full-Year 2022 Financial Results

Company maintains priority focus on clinical programs to develop a novel functional curefor hepatitis B viral (HBV) infection and a potential first-of-its-kind treatment forpostpartum depression (PPD) and major depressive disorders (MDD)
Multiple Phase 2a proof-of-concept (POC) clinical data readouts and Phase 2b clinical trial initiations expected in 2023
Operations well-funded through 2025
Company to host conference call today at 8:00 PM HKT / 8:00 AM ET
DURHAM,N.C. and BEIJING,March 24,2023 -- Brii Biosciences Limited ("Brii Bio," "we," or the "Company",stock code: 2137.HK),a biotechnology company developing therapies to improve patient health and choice across diseases with high unmet needs,today announced a corporate update and reported its annual results for the year ended December 31,2022.
"In 2022 we achieved several key clinical,commercial and corporate milestones that strengthen our overall position to tackle major public health challenges on behalf of patients." Stated Zhi Hong,Ph.D.,Chairman and Chief Executive Officer of Brii Bio,"Throughout the upcoming year,we will continue to advance our lead clinical development programs as we work to bring a functional cure for hepatitis B viral infection to patients in China and potential first-of-its-kind treatment options for postpartum depression and major depressive disorders to patients in the U.S. Across our broader infectious and central nervous system disease portfolios,we look forward to adding new best-in-class partnerships and applying our proven internal discovery capabilities to strategically shape and position the Company for long-term growth and success."
Beyond its lead clinical programs in hepatitis B viral (HBV) infection,postpartum depression (PPD) and major depressive disorders (MDD),Brii Bio and its partners are advancing a broad pipeline of more than 10 differentiated treatment options for anxiety and major depressive disorders,human immunodeficiency virus (HIV),multidrug resistant (MDR) and extensively drug resistant (XDR) Gram-negative infections and non-tuberculous mycobacterial (NTM) lung disease.
In addition,Brii Bio has made the decision to discontinue its amubarvimab/romlusevimab antibody combination program for COVID-19 and has stopped manufacturing efforts in order to redirect resources to high-priority programs. This determination is based on constantly evolving COVID-19 trends and policy updates,as well as protracted regulatory inspections at our contract development and manufacturing organization (CDMO) sites.
Fiscal Year 2022 and Recent Corporate Developments
Brii Bio expanded its executive leadership team and strengthened its Board structure and corporate development initiatives with the additions of Dr. Ankang Li,Chief Strategy and Financial Officer,to the Board of Directors as an executive director and chair of the Strategy Committee,and Dr. Taiyin Yang as an independent non-executive director of the Board and co-chair of the Audit and Risk Committee. New additions to the Brii Bio senior leadership team included Dr. Susannah Cantrell,Chief Business Officer,Dr. Eleanor de Groot,Chief Technology Officer,Dr. Aleksandar Skuban,Central Nervous System Diseases Therapy Area Head,and Karen D. Neuendorff,Chief People Officer.
Brii Bio strengthened the China leadership team,which included the appointments of Dr. Qing Zhu to Head of China Research & Development,and Mr. Rico Liang to General Manager of China.
Brii Bio was added to the MSCI China Small Cap Index,an international benchmark for global institutional investors seeking to optimize their investment portfolios.
Brii Bio commercially launched the amubarvimab/romlusevimab combination as a long-acting COVID-19 neutralizing antibody in China.
Brii Bio continued to foster partnerships with key maternal health advocacy groups to address patient needs and preferences in the U.S.,including supporting the Postpartum Support International's Climb Out of the Darkness event,and sponsorship of the 20/20 Mom Annual Forum,Maternal Mental Health Now,the 35th Annual Postpartum Support International Conference and the 2022 Black Maternal & Mental Health Summit.
Brii Bio continues to receive broad industry recognition for its corporate and clinical development accomplishments across more than 10 awards and feature lists,including "50 Women in Tech" by Forbes China,"2022 Annual Biotechnology Innovation" by China Times,"Top 10 Chinese Pharmaceutical Listed Companies in ESG Investment Value in 2022" by Healthcare Executives,"2022-2023 Gold Bell Seal for Workplace Mental Health" by Mental Health America and more. Brii Bio also received an "A" rating by MSCI ESG Rating,a globally recognized assessment of a company's resilience to long-term environmental,social and governance (ESG) risks.
Fiscal Year 2022 Clinical Pipeline Highlights and Upcoming Milestones
Hepatitis B Virus (HBV) Program (China team core project)
Led by Brii Bio's team in China and partners,Vir Biotechnology,Inc. ("Vir," NASDAQ: VIR) and VBI Vaccines,Inc. ("VBI," NASDAQ: VBIV),the Company is progressing multiple combination studies for the treatment of HBV to improve the probability of achieving a high rate of functional cure for chronic HBV patients in China.China has the largest prevalence of HBV in the world,with 87 million people impacted by this disease,yet there is no effective functional cure currently available for patients.
BRII-179 (VBI-2601) in Combination with BRII-835 (VIR-2218) (Study conducted by Brii Bio)
In February 2023,interim results were presented in an oral session at the Asian Pacific Association for the Study of Liver (APASL) 2023 indicating that combination therapy with BRII-835 (VIR-2218) and BRII-179 (VBI-2601) was safe and well-tolerated,induced stronger anti-hepatitis B surface antigen (HBsAg) antibody responses and led to improved HBsAg-specific T-cell responses,when compared with BRII-835 (VIR-2218) or BRII-179 (VBI-2601) alone. In the data presented at APASL,50 participants in all cohorts achieved HBsAg reduction at the end of treatment with a mean decrease of -1.7 to -1.8 log10 IU/mL. In addition,two participants in combination cohorts achieved maximum reductions in HBsAg at or below the lower limit of quantification (LLOQ) by Week 40,along with robust HBsAg-specific antibody and T-cell responses.
Additional data from the Phase 2 study of BRII-179/BRII-835 combination are expected in the second half of 2023.
BRII-179 (VBI-2601) in Combination with PEG-IFN-α (Study conducted by Brii Bio)
In December 2022,the Company completed patient enrollment in part one of a Phase 2 combination trial evaluating the addition of BRII-179 (VBI-2601) in chronic HBV patients already receiving pegylated interferon alpha (PEG-IFN-α) and nucleotide/nucleoside reverse transcriptase inhibitors (NRTI) treatment.
Topline results are expected in the second half of 2023.
VIR-2218 (BRII-835) in Combination with PEG-IFN-α (Study conducted by Vir)
Vir announced end-of-treatment data from an ongoing Phase 2 trial of combination 48 weeks of VIR-2218 (BRII-835) with PEG-IFN-α at the American Association for the Study of Liver Diseases (AASLD) 2022 meeting,which demonstrated that nearly 31% of patients with chronic HBV infection achieved HBsAg seroclearance with anti-HBs seroconversion with no new safety signals.
Additional data from the Phase 2 study led by Vir are expected in the first half of 2023.
VIR-2218 (BRII-835) in Combination with VIR-3434 (BRII-877) (MARCH Study conducted by Vir)
In November 2022,Vir presented initial end-of-treatment data at AASLD's The Liver Meeting® from Part A of its ongoing Phase 2 MARCH study evaluating VIR-2218 (BRII-835) in combination with VIR-3434 (BRII-877) in participants with chronic HBV infection who received NRTI therapy. The data indicated additive HBsAg reductions from VIR-2218 (BRII-835) and VIR-3434 (BRII-877) with combination regimens achieving a much greater HBsAg reduction than either alone in all patients with no safety signals.
Additional data from Part A of Vir's ongoing Phase 2 MARCH trial are expected in the first half of 2023.
Initial data to evaluate VIR-2218 (BRII-835) and VIR-3434 (BRII-877) with or without PEG-IFN-α,are expected in the second half of 2023 from Part B of Vir's ongoing Phase 2 MARCH trial.
Brii Bio is working closely with the Center for Drug Evaluation (CDE) of the National Medical Products Administration (NMPA) to initiate a Phase 1 study of BRII-877 (VIR-3434) in China.
Postpartum Depression (PPD) Program and Major Depressive Disorder (MDD) (U.S. team core project)
Brii Bio is developing its internally-discovered BRII-296 therapeutic candidate as a first-of-its-kind one-time injection treatment with the potential to expand the PPD and MDD treatment landscapes for patients in the U.S.
BRII-296
In September 2022,Brii Bio announced positive topline results from its Phase 1 study of BRII-296 with data that demonstrated a single administration of the investigational therapy at 600 mg delivered a favorable pharmacokinetic (PK) profile and was safe and well-tolerated in healthy subjects. Early feedback from physicians and patient communities is very positive and reinforces the potential for a first-of-its-kind single-injection treatment option for PPD.
Brii Bio is working closely with the U.S. Food and Drug Administration (FDA) to align and agree on a PPD treatment protocol in preparation for its Phase 2 proof-of-concept (POC) study.
The Company is actively working to expand the clinical indications for BRII-296 in 2023 and plans to initiate additional Phase 2 studies in the U.S. by the end of the year.
BRII-297
Brii Bio has conducted Investigational New Drug (IND)-enabling studies with BRII-297 targeting various anxiety and depressive disorders.
Brii Bio plans to initiate a first-in-human PK,safety and tolerability study with BRII-297 in Australia in the first half of 2023.
Additional Clinical and Pre-Clinical Development Updates
Human Immunodeficiency Virus (HIV) Program
BRII-732
In October 2022,Brii Bio presented positive Phase 1 data showing that BRII-732 demonstrated an acceptable safety and tolerability profile,as well as a favorable and linear PK profile that achieved therapeutic targets in healthy volunteers,reinforcing its potential as an oral once-weekly therapy for the treatment of HIV infections.
In December 2022,Brii Bio was notified by the U.S. FDA that it had lifted the clinical hold on the Company's planned Phase 1 study to investigate a lower oral dose of once-weekly BRII-732. The Company is exploring partnership opportunities to continue developing BRII-732 as part of a potential oral,once-weekly,long-acting combination treatment option for HIV patients.
BRII-753
The Company also selected a new clinical candidate,BRII-753,as a long-acting subcutaneous injection therapy with the goal to extend the dosing schedule to once monthly,once quarterly or twice-yearly.
BRII-778
Based on the PK data from a completed Phase 1 study,which determined additional development work was required to achieve optimal PK targets for the treatment of HIV,Brii Bio has made the decision to discontinue development of BRII-778.
Multidrug- and Extensively Drug-Resistant (MDR/XDR) Gram-negative Bacteria Infections Program
BRII-693 (QPX-9003)
Qpex Biopharma,Inc. (Qpex) announced in early 2022 that BRII-693 received Qualified Infectious Disease Product (QIDP) designation by the U.S. FDA.
In October 2022,Qpex presented at IDWeek interim Phase 1 results from its completed first-in-human clinical study,demonstrating that BRII-693 is safe and well-tolerated at all doses tested and supports continued development of BRII-693 for the treatment of Acinetobacter baumannii and Pseudomonas aeruginosa infections resistant to carbapenem.
Qpex continues to work closely with the U.S. FDA to align its next steps in clinical development.
Brii Bio plans to submit a pre-IND to the NMPA in the first half of 2023 for development of BRII-693 in China.
BRII-672 (ORAvance™)
Qpex announced in early 2022 that BRII-672,in combination with a non-disclosed oral beta-lactam antibiotic,received QIDP designation by the U.S. FDA.
Preclinical data and interim Phase 1 clinical results were presented at IDWeek in October 2022.
In the fourth quarter of 2022,Qpex completed the first-in-human Phase 1 study in the U.S. No subjects discontinued treatment due to adverse events (AEs) and no serious adverse events (SAEs) were observed in this Phase 1 single ascending dose (SAD) study.
In December,Brii Bio submitted a pre-IND to the NMPA seeking regulatory guidance around a development plan for BRII-672 in China.
Qpex continues to work closely with the U.S. FDA to align its next steps in clinical development.
BRII-636 (OMNIvance®)
Qpex announced in early 2022 that BRII-636 received QIDP designation by the U.S. FDA.
Qpex completed a first-in-human Phase 1 study and a drug-drug interaction study and presented findings at IDWeek in the fourth quarter of 2022. This Phase 1 multiple ascending dose (MAD) study indicated that overall,BRII-636 (xeruborbactam),alone and in combination with meropenem,at doses associated with efficacy in animal models of infection was well-tolerated.
Qpex continues to work closely with the U.S. FDA to align its next stages in clinical development.
Non-tuberculous Mycobacterial (NTM) Lung Disease Program
BRII-658 (epetraborole)
Brii Bio's partner,AN2 Therapeutics,Inc. (NASDAQ: ANTX),is advancing a pivotal Phase 2/3 clinical trial for treatment-refractory Mycobacterium avium complex (MAC) lung disease.
AN2 also completed and reported topline results from its Phase 1 bridging study designed to evaluate the PK,safety and tolerability of oral BRII-658 (epetraborole) in Japanese subjects.
COVID-19 Program
Following commercial launch in July 2022,Brii Bio sold substantially all available products of the amubarvimab/romlusevimab combination,with distribution to 25 provinces and 358 hospitals,with a revenue of RMB51.6 million. And as part of its commitment to ensuring humanitarian access and contributing to the containment of the pandemic outbreak,the Company donated nearly 3,000 doses for emergency use to 21 cities and 22 hospitals in China prior to the commercial launch.
In January 2023,the amubarvimab/romlusevimab combination is the recommended antiviral treatment for COVID-19 in both the 10th COVID-19 Diagnosis and Treatment Guideline and the 4th COVID-19 Diagnosis and Treatment Protocol for Severe/Critical Cases.
The Company has made the decision to discontinue its amubarvimab/romlusevimab antibody combination program and has stopped manufacturing efforts in order to redirect resources to high-priority programs. This determination is based on the constantly evolving COVID-19 trends including the upcoming expiration of the federal Public Health Emergency (PHE) by the U.S. Department of Health and Human Services' (HHS) in May 2023,as well as protracted regulatory inspections at our CDMO sites. The Company is working with the U.S. FDA to withdraw the Emergency Use Authorization (EUA) application at an appropriate time following the completion of activities required by the regulatory authority and also with the China NMPA to withdraw the Biologics License Application (BLA) in the third quarter of 2023 once all necessary regulatory requirements have been completed. No significant revenue is expected in the future from the commercialization of amubarvimab/romlusevimab injection combination either in China or in the U.S. and other territories.
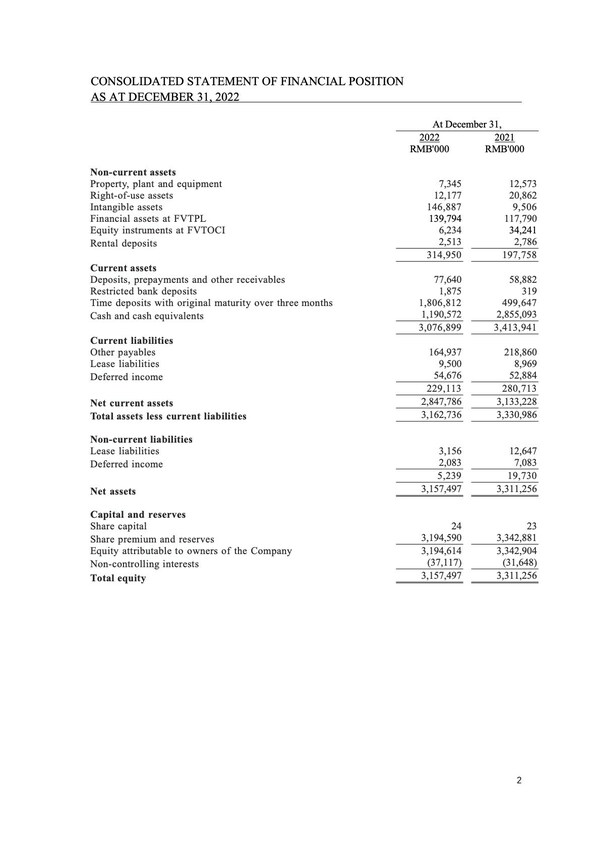
Full Year 2022 Financial Results
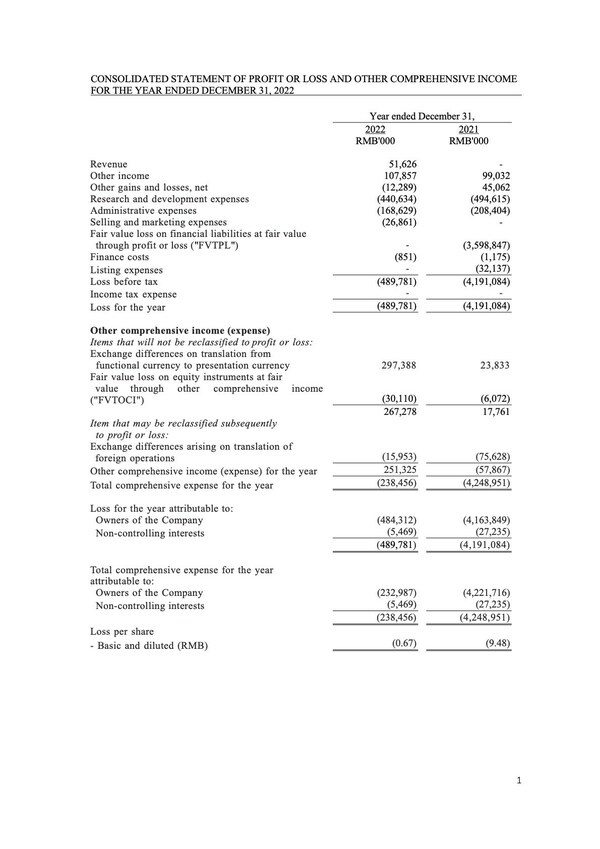
The revenues increased by RMB51.6 million from nil for the year ended December 31,2022. The increase was due to the commercialization of the long-acting amubarvimab/romlusevimab combination therapy in China for the treatment of COVID-19.
Other income was RMB107.9 million for the year ended December 31,2022,representing an increase of RMB8.9 million or 9.0%,compared with RMB99.0 million for the year ended December 31,2021. The increase was mainly due to the increase in bank interest income of RMB30.7 million attributable to the increased bank and cash balances after the Global Offering. The increase was partially offset by the decrease in income recognized from PRC government grants.
Research and development expenses were RMB440.6 million for the year ended December 31,representing a decrease of RMB54.0 million,or 10.9%,compared with RMB494.6 million for the year ended December 31,2021. The decrease was primarily due to the decrease in third party contracting costs relating to COVID-19 programs. It was partially offset by the increase in the employee cost for our continuous development in clinical trials.
Selling and marketing expenses increased by RMB26.9 million from nil for the year ended December 31,2022. The increase was primarily attributable to the commercialization of COVID-19 therapy.
Administrative expenses were RMB168.6 million for the year ended December 31,representing a decrease of RMB39.8 million,or 19.1%,compared with RMB208.4 million for the year ended December 31,2021. The decrease was primarily attributable to the decrease in the employee costs.
Total comprehensive expense for the year ended December 31,2022 was RMB238.5 million,representing a decrease of RMB4,010.5 million,or 94.4%,compared with RMB4,249.0 million for the year ended December 31,2021. The decrease was primarily attributable to the decrease in fair value loss on financial liabilities at FVTPL.
Conference Call Information
A live conference call will be hosted on March 24,2023,at 8:00 PM Hong Kong time (8:00 AM U.S. Eastern Time). All participants are required to register in advance of the call. For the registration link,please click here.
All participants shall use the link provided above to complete the online registration process in advance of the conference call. Upon registering,each participant will receive an email with important details for this call,including the call date,time and access link. This link is to be kept confidential and not shared with other participants. Additionally,a replay of the conference call will be available after the call and can be accessed by visiting the Company's website at www.briibio.com under the Investor Relations section.
About Brii Bio's Programs
Hepatitis B Virus (HBV) (Licensed from VBI and Vir,China team core project)
As one of its leading clinical development programs,Brii Bio is building a broad pipeline of novel HBV therapeutic candidates in order to improve the probability of achieving a high rate of functional cure for each subpopulation of HBV patients. Each of Brii Bio's HBV candidates has a unique therapeutic modality with proven clinical benefit targeting this chronic infection,which allows the Company to explore an expansive set of potential combination treatment options for various patient subgroups. Brii Bio holds exclusive rights in Greater China to develop and commercialize BRII-179 (VBI-2601),BRII-835 (VIR-2218) and BRII-877 (VIR-3434).
BRII-179 (VBI-2601)is a novel recombinant protein-based HBV immunotherapeutic candidate that expresses the Pre-S1,Pre-S2,and S HBV surface antigens,and is designed to induce enhanced B-cell and T-cell immunity.
BRII-835 (VIR-2218)is a GalNAc-conjugated small interfering ribonucleic acid (siRNA) targeting all HBV viral RNAs that has shown to block viral transcription,reduce viral protein and alleviate immune suppression.
BRII-877 (VIR-3434)is an investigational subcutaneously administered HBV-neutralizing monoclonal antibody designed to block entry of all 10 genotypes of HBV into hepatocytes and also to reduce the level of virions and subviral particles in the blood. BRII-877 (VIR-3434),which incorporates Xencor's Xtend™ and other Fc technologies,has been engineered to potentially function as a T cell vaccine against HBV in infected patients,as well as to have an extended half-life.
Postpartum Depression (PPD) and Major Depressive Disorder (MDD)/Other CNS disorders (Internally discovered,U.S. team core project)
Leveraging patient insights,Brii Bio is developing BRII-296 and BRII-297 to expand treatment options for patients with psychiatric disorders who are often underserved and overlooked across the industry. Utilizing applied drug formulation know-how to develop long-acting therapies,Brii Bio is focused on improving drug administration convenience and patient compliance to ensure potential treatment success.
BRII-296is a novel,long-acting,single injection therapeutic candidate in development for the treatment PPD and MDD. It acts as a gamma-aminobutyric acid A (GABAA) receptor positive allosteric modulator (PAM) and is designed to provide a rapid,profound and sustained reduction in depressive symptoms of PPD and MDD with the potential to lead to greater adherence,convenience and fewer side effects compared to the current standard of care.
BRII-297is a new chemical entity (NCE) discovered internally in development as a long-acting injectable (LAI) treatment of various anxiety and depression disorders.
Human Immunodeficiency Virus (HIV) (Internally discovered,U.S. team project)
Brii Bio is seeking partners to collaborate on the development of BRII-732 as a once-weekly oral single-tablet regimen for the treatment or prevention of HIV. Brii is also seeking development partnership for BRII 753,a novel low volume,subcutaneous injection therapy with potential to dose monthly to every six months. Both compounds demonstrate considerable promise to serve as a key component for long-acting HIV treatment regimens that will offer more discreet and convenient options for patients living with HIV,and as monotherapy for HIV prevention.
BRII-732is a proprietary prodrug NCE that,upon oral administration,is rapidly metabolized into EFdA and is under evaluation as a potential HIV treatment or prevention option. BRII-732 is a nucleoside analogue reverse transcriptase translocation inhibitor (NRTTI) that acts as both a chain terminator and translocation inhibitor of HIV.
BRII-753is an NCE currently in the preclinical stage of development. It has been internally discovered and is being developed as a long-acting subcutaneous injection with potential for dosing once monthly to once every six months. BRII-753 can be used in a combination therapy for HIV treatment and as monotherapy for pre-exposure prophylaxis (PrEP).
Multidrug- and Extensively Drug-Resistant (MDR/XDR) Gram-negative Infections (Licensed from Qpex,China team project)
Brii Bio is developing MDR/XDR therapies in collaboration with Qpex as part of their global development plan. Based on a licensing agreement with Qpex,Brii Bio has the exclusive rights to develop and commercialize BRII-636,BRII-672 and BRII-693 in Greater China. Qpex is progressing BRII-636,BRII-672 and BRII-693 in parallel with a goal of moving each to global Phase 3 studies,at which time Brii Bio will participate in the China-based arm of the global research protocols. BRII-636,BRII-672 and BRII-693 candidates all obtained QIDP designation from the U.S. FDA,which may receive incentives in the future.
BRII-693 (QPX-9003)is a novel synthetic lipopeptide in development for the treatment of MDR/XDR Gram-negative bacterial infections. Based on a combination of increased in vitro and in vivo potency,and an improved safety profile compared with currently available polymyxins,BRII-693 has the potential to be an important addition to hospital-administered intravenous antibiotics.
BRII-672 (ORAvanceTM) is a prodrug of BRII-636 and an oral beta-lactamase inhibitor (BLI) in development for the treatment of MDR/XDR Gram-negative bacterial infections. These agents were discovered by Qpex as part of their expertise in BLIs,using the boron atom as a part of its pharmacophore.
BRII-636(OMNIvance®) is an intravenously administered novel cyclic boronic acid derived broad-spectrum inhibitor in development for the treatment of MDR/XDR Gram-negative bacterial infections.
Non-tuberculous Mycobacterial (NTM) Lung Disease Program (Licensed from AN2,China team project)
Brii Bio's strategic partner,is developing epetraborole (BRII-658) as a once-daily oral treatment for patients with chronic NTM lung disease,with an initial focus on treatment-refractory Mycobacterium avium complex (MAC) lung disease,which is the subpopulation of MAC lung disease with the highest unmet medical need for new therapies. Brii Bio holds a license to develop,manufacture and commercialize epetraborole (BRII-658) in Greater China.
BRII-658 (epetraborole)is in development as a once-daily oral treatment for patients with chronic NTM lung disease,with an initial focus on treatment of refractory MAC lung disease. It is a boron-containing,small molecule inhibitor of mycobacterial leucyl-tRNA synthetase,or LeuRS,an enzyme that inhibits protein synthesis.
COVID-19 (Discovered in collaboration with Tsinghua University and Third People's Hospital of Shenzhen through Brii Bio's subsidiary,TSB Therapeutics Ltd (Beijing) Co. Limited.)
Amubarvimab and romlusevimab are non-competing SARS-CoV-2 monoclonal neutralizing antibodies derived from convalesced COVID-19 patients. They have been specifically engineered to reduce the risk of antibody-dependent enhancement and prolong the plasma half-lives for potentially more durable treatment effect.
Approved by China's NMPA in December 2021,the long-acting amubarvimab/romlusevimab cocktail therapy is approved to be administered by intravenous infusion in two sequential doses for the treatment in adults and pediatric patients (age 12-17 weighing at least 40 kg) of mild- and normal-type COVID-19 at high risk for progression to severe disease,including hospitalization or death. The indication of pediatric patients (age 12-17 weighing at least 40 kg) is under conditional approval. In January 2023,the National Health Commission of China reiterated the amubarvimab/romlusevimab combination in its COVID-19 Diagnosis and Treatment Guidelines (10thEdition) for the treatment of COVID-19 and the 4th COVID-19 Diagnosis and Treatment Protocol for Severe/Critical Cases. The live virus testing data as well as pseudovirus testing data from multiple independent labs have demonstrated that the amubarvimab/romlusevimab combination retains activity against commonly identified SARS-CoV-2 variants B.1.1.7 (Alpha),B.1.351 (Beta),P.1 (Gamma),B.1.429 (Epsilon),B.1.617.2 (Delta),AY.4.2 (Delta Plus),C.37 (Lambda),B.1.621 (Mu),B.1.1.529-BA.1 (Omicron) and BA.1.1,BA.2,BA.2.12.1,BA.4/5,BF.7 (Omicron subvariants).
This press release contains references to third-party information. Such information is not deemed to be incorporated by reference in this press release. Brii Bio disclaims responsibility for such third-party information.
About Brii Bio
Brii Biosciences ("Brii Bio",stock code: 2137.HK) is a biotechnology company developing therapies to address some of the world's most common diseases where patients experience high unmet medical needs,limited choice and significant social stigmas. With a focus on infectious and central nervous system diseases,the Company is advancing a broad pipeline of unique therapeutic candidates with lead programs to develop a novel functional cure for hepatitis B viral infection (HBV) and a first-of-its-kind treatment for postpartum depression (PPD). The Company is led by a visionary and experienced leadership team and has operations in key biotech hubs,including Raleigh-Durham,the San Francisco Bay Area,Beijing and Shanghai. For more information,visit www.briibio.com.
Forward Looking Statement
The information communicated in this press release contains certain statements that are or may be forward looking. These statements typically contain words such as "will," "expects," "believes," "plans" and "anticipates," and words of similar import. By their nature,forward looking statements involve risk and uncertainty because they relate to events and depend on circumstances that will occur in the future. There may be additional material risks that are currently not considered to be material or of which the Company are unaware. These forward-looking statements are not a guarantee of future performance. Against the background of these uncertainties,readers should not rely on these forward-looking statements. The Company assumes no responsibility to update forward-looking statements or to adapt them to future events or developments.
Tags: Banking/Financial Service Biotechnology Health Care/Hospital Infectious Disease Control Medical/Pharmaceuticals Pharmaceuticals
Previous:World TB Day 2023: Illumina and GenoScreen Launch Next Generation Sequencing Inn...
Next:Clarivate Announces Gordon Samson as President, Intellectual Property and Nomina...
Leave a comment
Follow Us
Newsletter
Join us to get the latest news.




