iSTAR Medical receives U.S. FDA approval to start pivotal trial for MINIject in glaucoma patients

WAVRE,Belgium,July 15,2021 --iSTAR Medical,a med-tech company developing minimally-invasive implants for glaucoma surgery (MIGS),announced today that the U.S. Food and Drug Administration (FDA) granted it Investigational Device Exemption (IDE) to start a pivotal trial with MINIjectTM. The STAR-V study will investigate MINIjectTM in over 350 patients with primary open angle glaucoma. World-leading glaucoma surgeons in the US,Canada and Europe will join the trial.
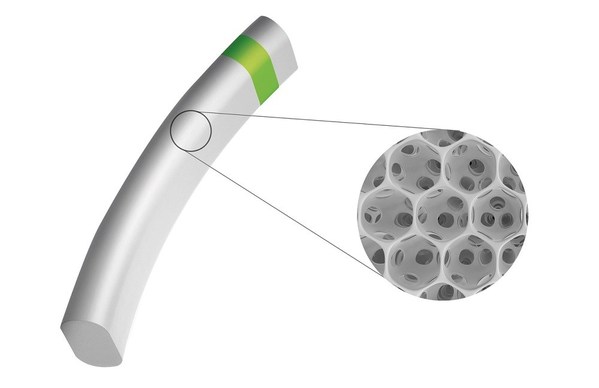
MINIject implant made of proprietary STAR material
The STAR-V trial evaluates MINIject's efficacy by the mean reduction in eye pressure,as well as the proportion of patients achieving at least a 20 percent reduction in eye pressure. This study will report on safety and efficacy of MINIject alone,in a procedure not combined with simultaneous cataract surgery. Key study findings will become available when all patients have completed 2 years in the study. Patients will also be followed to evaluate long-term benefits and tolerability of MINIjectTM in the treatment of mild to moderate glaucoma.
Dr. Brian E. Flowers,a glaucoma specialist at Ophthalmology Associates of Fort Worth,TX,USA said "On behalf of the investigators,I would like to share our excitement to start the STAR-V trial. The supraciliary space of the eye is a natural outflow pathway which has demonstrated great promise. There is the potential to deliver increased efficacy in lowering eye pressure for our patients in a less invasive fashion. Access to an effective pressure lowering device that is used in a standalone procedure will enable us to offer treatment to many more glaucoma patients with a minimally-invasive treatment option."
Michel Vanbrabant,CEO of iSTAR Medical,commented "We are very pleased that the FDA has granted us approval to bring this innovative technology to North American patients suffering from primary open angle glaucoma in the STAR-V trial. Results from clinical trials in over 130 patients in Europe,Asia and Latin America have consistently demonstrated that MINIjectTM maintains a positive safety profile,and delivers a significant reduction of pressure thanks to our proprietary STAR material and the power of the supraciliary space."
About iSTAR Medical
iSTAR Medical SA is a private med-tech company developing minimally invasive ophthalmic implants for the treatment of glaucoma patients. iSTAR Medical has exclusive rights for ophthalmic use of the STAR® material,developed by the University of Washington,Seattle (USA). STAR has outstanding anti-fibrotic and anti-inflammatory properties and a unique porous structure that enhances natural fluid outflow. iSTAR Medical was founded in 2011 and is headquartered in Wavre,Belgium. www.istarmed.com
About MINIjectTM
MINIjectis iSTAR Medical's revolutionary MIGS device for patients with primary open-angle glaucoma. MINIject combines the unique porous structure of its proprietary STAR material with the power offered by the supraciliary space. As a result,it is designed to enhance natural fluid outflow,reducing intraocular pressure (IOP) and the need for medication,while bio-integrating with surrounding tissue,limiting inflammation,fibrosis and subsequent complications.
About Glaucoma
Glaucomais a progressive disease and the second leading cause of adult blindness,1 affecting over 100 million people globally.2 IOP reduction,through medication or surgery,helps delay disease progression.1 Medication is generally the first line treatment,but the progressive addition of multiple drops can burden patients with side effects,compliance challenges and costs.1,2 Invasive surgery can present risks with irreversible complications.1,2 MIGS is the most promising and fastest-growing glaucoma therapy due to its enhanced safety profile.2 MINIject is potentially best-in-class for its promising long-term efficacy and safety.
[1]"European Glaucoma Society Terminology and Guidelines for Glaucoma",4th Edition: British Journal of Ophthalmology. 2017;101:1-195 https://bjo.bmj.com/content/101/5/73
[2]"Market Scope Glaucoma Surgical Device Market Report",August 2020. https://www.market-scope.com/pages/reports/202/2020-glaucoma-surgical-device-market-report-a-global-analysis-for-2019-to-2025-august-2020

View original content to download multimedia:https://www.prnewswire.com/news-releases/istar-medical-receives-us-fda-approval-to-start-pivotal-trial-for-miniject-in-glaucoma-patients-301333291.html